Chemistry Solutions
May 2026 | Tech Tips
Computational Chemistry in the High School Classroom Exploring a dynamic way to help students build their chemistry interest, confidence and understanding
By Michael Bowman
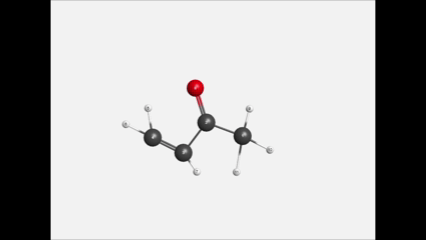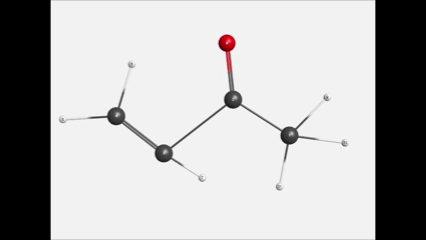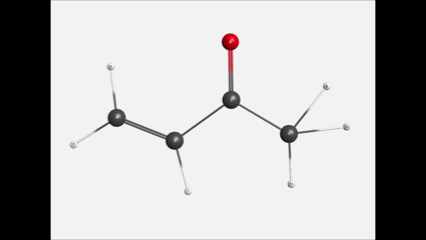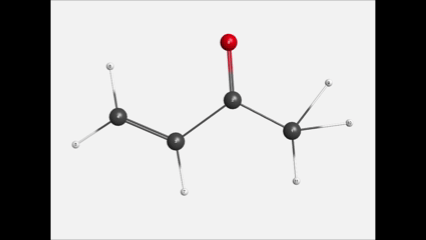 |
Figure1. Animation showing a geometric optimization of C4H6O, created using computational chemistry on the WebMO demo server. |
Computational chemistry (CC) is the branch of chemistry that employs mathematical models and computer programs to analyze chemical systems. Over the last five decades, the processing power of computers has grown exponentially, which has vastly expanded the capacity of computational methods in chemistry. Now computational chemistry appears in virtually all areas of modern chemical research. Over the last 30 years, three Nobel prizes have been awarded to breakthroughs in computational chemistry.
Amazingly, some of the same computational methods used by professionals can be used by your students to help them learn a wide variety of concepts about thermochemistry, atomic orbitals, or VSEPR theory — at little to no cost to your school. Moreover, numerous studies1,2,3,4 have demonstrated the potential impact of incorporating computational chemistry into chemistry classes, including increasing student interest, confidence, and knowledge.
While many college instructors have adopted CC methods into chemistry classrooms and labs, it appears that high schools have lagged in this regard. Many high school teachers may be unaware of the nature of computational chemistry, while others may have fundamentally misunderstood the role it can play inside the classroom. Below I’ll address three common misconceptions that may have discouraged high school chemistry teachers from using computational chemistry in their classrooms and then illustrate how I’ve implemented CC methods into my own courses.
Misconception #1: “Computational chemistry is just another visual tool”
One of the main struggles for students as they learn chemistry is imagining the submicroscopic realm of atoms and molecules. In response, chemistry instructors have employed a wide variety of tools to help students visualize chemical phenomena, including balloon art, molecular modeling kits, 3D printed models, and virtual reality. Computational chemistry methods likewise can display 3D views of molecules and orbitals, but these programs go far beyond serving as mere visual aids. With computational chemistry, students can use a graphical user interface (GUI), such as WebMO or Gaussview, to easily interact with a variety of quantum chemistry software packages to construct their own molecules, one atom at a time, and to calculate properties as they please (see Figure 1).
For instance, a student can build a water molecule to visualize its dipole moment, then replace a hydrogen atom with fluorine and recompute to see how the dipole moment is affected by the change (Figure 2). The possibilities are limitless. This freedom promotes curiosity in a manner that is difficult to replicate with other chemistry activities. Recent research5
has revealed a strong correlation between chemistry students’ curiosity and their motivation and interest. Considering this connection, it’s natural to expect that computational chemistry strategies can be used to inspire students to thoughtfully engage with complicated concepts in chemistry, even without understanding the associated math that makes it work.
Misconception #2: “Computational chemistry is too complicated for my students and for me”
Computational chemistry often involves cryptic acronyms like “B3LYP” or “6-311++G(2d,p)” and, up to now, few chemistry educators have received college training on computational methods. As a result, many educators feel too intimidated to use these methods. A recent study by Traube and Blonder6 found that many high school teachers lack confidence in implementing computational chemistry in their classrooms, even after completing an 18-hour course on the matter. Even if you comfortably understand computational chemistry methods, you may sense this topic is too advanced for your students. However, I believe teachers only need to know and communicate the fundamentals of computational chemistry (Table 1) to successfully implement these activities into their curricula.
Computational Chemistry: the 5 Ws |
|
Who? |
|
What? |
|
When? |
|
Where? |
|
Why? |
|
Table 1. A high-level overview of computational chemistry. |
Teachers can introduce the principles of CC by referring to fundamental concepts, like atomic properties and Coulomb’s Law, to convince students that the properties of atoms and molecules are governed by their components. Students can then imagine how the behavior of electrons and nuclei can be explained by advanced mathematical equations. While these equations are exact in theory, solving them by hand — even for a two-electron system like helium — is impossible. Considering the vast number of interactions between subatomic particles in even a modestly sized system will quickly lead a student to recognize why high-performance computing is necessary to store and analyze the large amounts of data involved. Armed with this basic understanding, students can then treat the CC software as a ‘black box’ which transforms basic input into useful molecular properties such as energy and dipole moment. Fortunately, integrative GUIs such as WebMO make this process even simpler by allowing students to run computational chemistry jobs and view the results without struggling with the technical challenges of constructing input files or parsing output files. In my experience, most students can quickly grasp these fundamentals and some find CC activities to be more intuitive than other aspects of the course.
Misconception #3: “Computational chemistry is too expensive for my institution’s budget”
Modern computational chemistry research is often performed on high-performance servers that cost tens of thousands of dollars (even at the low-end). However, the computational resources required to perform the routine calculations suitable for an educational setting are quite minimal in comparison. Most teachers I know prefer to use one of the many free computational chemistry packages, such as Psi4, ORCA, or OpenMopac, along with free GUIs such as WebMO or Avogadro.
The lesson included with this article was written using the WebMO Demo Server, which is one of a few free remote-access online servers. The WebMO demo server runs the same major software packages as the paid, professional version, but limits the size and complexity of what can be analyzed. For those interested in pursuing CC concepts in greater depth, a server with as little as 8 GB of RAM would usually be sufficient to enable a class of 20 students to perform computational chemistry jobs.
What does computational chemistry look like in the classroom?
Incorporating computational chemistry requires only minor adjustments to how you teach your curriculum, and there’s considerable flexibility in how to do so. I find it effective to give brief CC demonstrations during lectures in order to maintain student engagement.
For instance, when discussing greenhouse gases in a general education course, I’ll compute the geometries and frequencies of a couple hydrofluorocarbons on WebMO during my lecture, and then show the class the simulated infrared spectra and play music while students watch the molecules moving in their various vibrational modes. This fun, simple activity helps students understand why the global warming potentials (GWPs) of atmospheric gases vary wildly. Altogether, this exercise takes no more than 10 minutes of class time. Later in the week, my students perform the same computational analysis for other greenhouse gases to explore molecular structure and vibrational frequencies in more depth during a 2-hour lab session.
Even though I teach at the college level, I’ve had the privilege to perform similar computational exercises in a high school classroom, with equal success, through the ACS Science Coaches program. In fact, with limited lab equipment and considerations of safety and prep time, I have found the barriers to implementing computational chemistry activities in a high school setting to be much lower than barriers to implementing traditional “wet” labs. If you are interested in trying computational chemistry in your classroom, you can begin with the downloadable activity on the side bar, which was designed specifically for high school students; or you can examine the free resources listed in Figure 3 below.
Where do I go to get started?The best way for beginners to gain experience with computational chemistry is to simply follow the instructions on an introductory activity, such as those curated at the following links: Fortunately, many of the activities on these sites are designed for high school students, and teachers may employ them directly in their classes. The author recommends prioritizing topics with strong visual components such as atomic orbitals, VSEPR theory, and polarity. Once you are familiar with the tools and methods in computational chemistry, you can also modify the provided instructions to align with the focus of your courses. |
Figure 3. Resources for teachers interested in incorporating computational chemistry into their lessons. |
In summary, computational chemistry is just as appropriate for the teaching of high school chemistry as it is for research at a top-tier research institution. It provides a vast array of pedagogical benefits and may be applied to a range of topics, from molecular orbital theory to thermodynamics or kinetics. The barriers that once restricted the use of computational chemistry have been significantly mitigated in recent decades. It is now possible to implement computational chemistry activities into your classroom without spending much of your time or budget. Try it for yourself, and you’ll see just how useful and easy computational chemistry can be.
References
1Hrubeš, J.; Jaroš, A.; Nemirovich, T.; Teplá, M.; Petrželová, S. Integrating Computational Chemistry into Secondary School Lessons. Journal of Chemical Education. 2024, 2343-2353.
2Sendlinger, S.; Metz, C. Computational Chemistry for Chemistry Educators. Journal of Computational Science Education. 2010, 28-32.
3Sharma, A. A Model Scientific Computing Course for Freshman. Journal of Computational Science Education. 2017, 2-9.
4Martin, N. Integration of Computational Chemistry into the Chemistry Curriculum. Journalp;l of Chemical Education. 1998, 241-243.
5Jannah, M.; Sutrisno, H. Study of the students’ curiosity relationship, interest and motivation in chemistry learning; a systematic literature review. AIP Conference Proceedings, Volume 2622, Yogyakarta, Indonesia, Nov 19-20, 2021; Retnowati, E.; Wiyarsi, A.; White, P.; Thoe, N.; Hamdi, S.; Fauzi, F.; Dwandaru, W., Eds.; AIP Publishing, Melville, NY, 2024.
6Traube, T.; Blonder, R. A Computational Chemistry Course for Teachers: From Research Laboratories to High-School Chemistry Teaching. Journal of Chemical Education. 2023, 4360-4368.

